Solution-focused toxicological risk assessment and medical device biocompatibility consulting
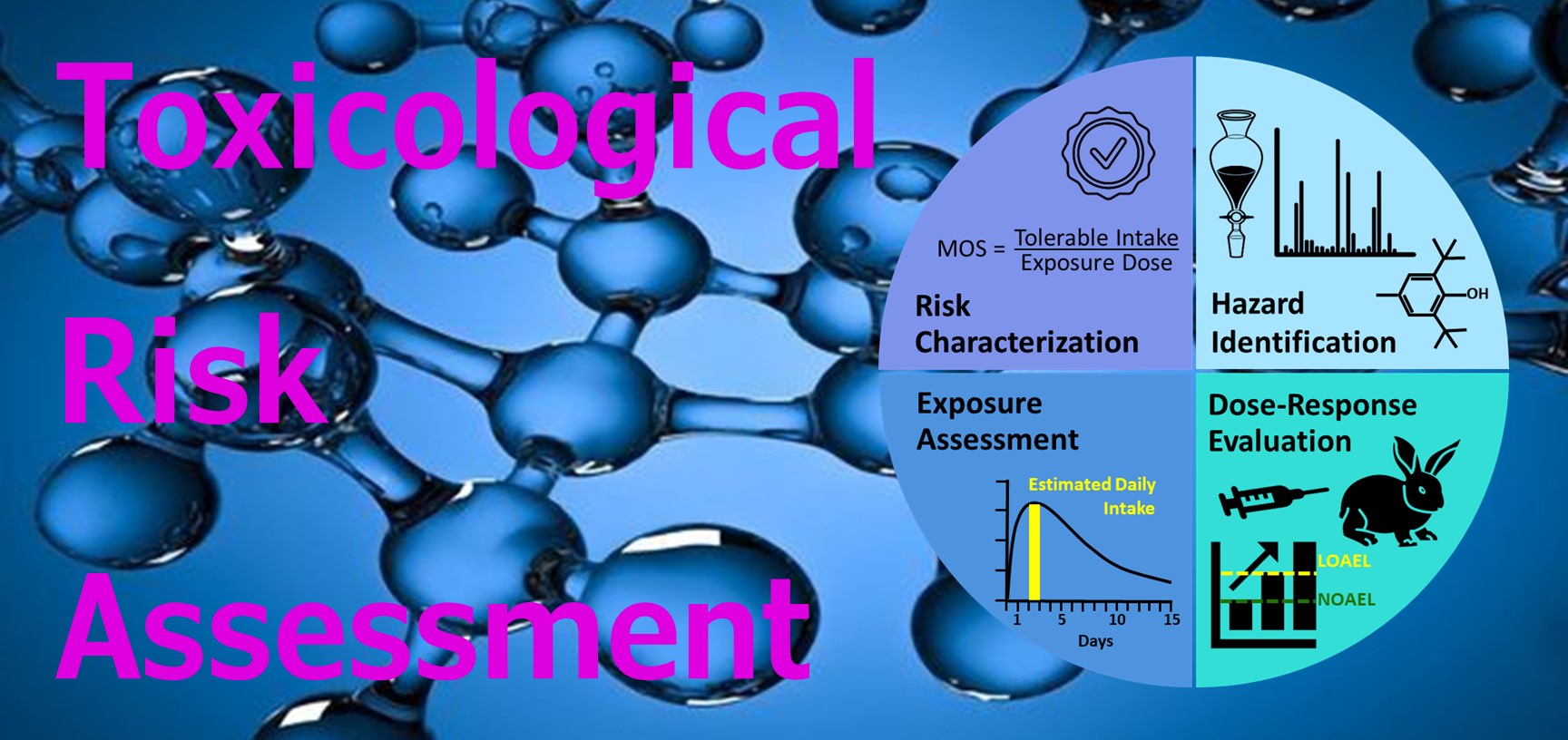
ISO compliant toxicological risk assessments (TRA)
· ISO 10993-17 TRA of medical devices (direct or indirect contact)
· ISO 18562 TRA of inhalation medical devices (gas flow pathway)
· ICH Q3E TRA of drug container closure systems and delivery devices
Assess toxicological risks of manufacturing nonconformances
Materials-based risk evaluation for mutagens or carcinogens
Quantitative risk assessment of potential sensitizers/irritants
As the author of more than 245 toxicological risk assessments (TRAs) and peer reviewer of 189 TRAs, TDAC has been consistently successful in obtaining 510(k) clearance of medical device submissions to US FDA, CE mark approval under EU MDR, and approvals by Japan PMDA, China, and other countries. TDAC has prepared TRAs for a wide variety of medical devices, including drug product container closure systems (CCS), prefilled syringes, drug coated stents, balloon catheters, vascular, ventricular, and urinary catheters, cardiac valves and artificial pumps, hemodialysis devices, in situ curing bone cements for hip and knee arthroplasty, in situ polymerizing cyanoacrylates for artificial embolic treatment of stroke, tissue adhesives, intravenous and subcutaneous drug delivery systems, transdermal patches, intraosseous needles, breast implants, wound dressings, tissue scaffolds, craniomaxillofacial reconstruction implants, orthopedic and dental implants, surgical instruments, bioresorbable sutures, 3-D printed bone scaffolds, humidified breathing circuits, MDI/DPI inhalers, nebulizers, oxygen and nitric oxide delivery systems, CPAP devices, and endotracheal and nasopharyngeal tubes.
TDAC’s board certified toxicologist (DABT) has expert knowledge of international and region-specific regulatory guidelines, including ISO 10993-1 through 10993-18; EU 2017/745, EU 722/2012, ISO 21726, ISO 14971, ISO 19227, ICH M7, Q3B, Q3C, Q3D, and Q3E; ISO 7405, ISO 18562, ISO 16000-6, USP 1663 and 1664; ASTM F1980-07; FDA guidelines for risk-benefit determination, wound dressings, tissue adhesives, surgical sutures, bone grafts, methacrylate bone cements, combination products, drug-eluting stents, breast implants, dental implants, MDI/DPI inhalers; SCHEER phthalate guidelines; and EMA 169430.
With over 28 years of experience in toxicological risk assessment, TDAC’s principal toxicologist has contributed toxicity profiles for 85 to 90% of chemicals assessed by a team of 10 toxicologists at one firm, as well as authored numerous profiles at other firms. Expertise in toxicological literature research stems from a familiarity with a multitude of governmental and industry databases from around the world, including USEPA IRIS, US CDC ATSDR, US EPA Provisional Peer Reviewed Toxicity Values (PPRTV), US EPA Pesticide Reregistration Status Reports, US DOE ORNL, California OEHHA, US Consumer Products Safety Commission, Joint FAO/WHO Expert Committee on Food Additives, European Chemicals Agency (ECHA) Registration Dossiers, ECHA Substance Information Data Set (SIDS) monographs, World Health Organization (WHO) monographs, IARC monographs, Cosmetic Ingredients Review (CIR) assessments, German Research Foundation MAK Collection, Human and Environmental Risk Assessment (HERA) chemical assessments, International Toxicity Estimates for Risk (ITER), Lhasa Carcinogenicity Potency Database (CPDB), Generally Recognized as Safe (GRAS) food additives database, occupational exposure limits (NIOSH, ACGIH, OSHA), National Research Council Recommended Daily Allowances (RDA), Institute of Medicine Dietary Reference Intakes (DRI) and Upper Level (UL) Intakes, Metabolomics database Foodb.ca, ICH Q3D, ICH M7, and PubChem Hazardous Substances Database (HSDB).
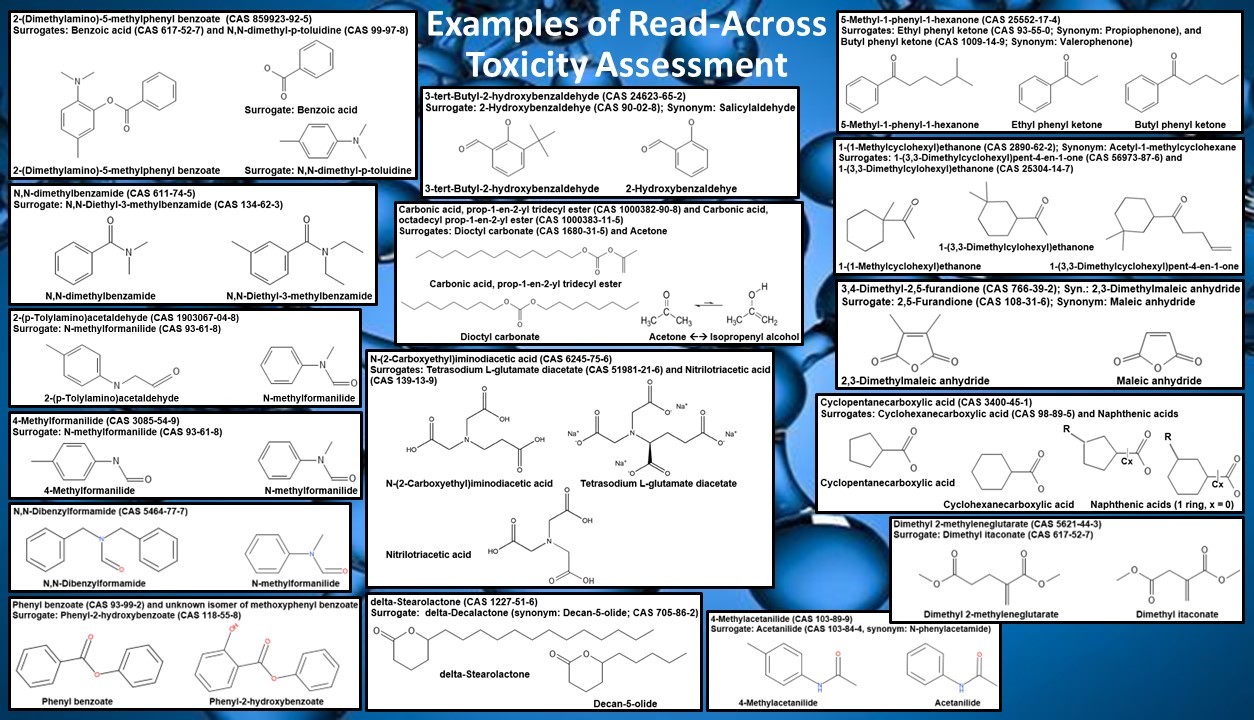
TDAC is able to identify surrogate compounds having similar chemical structures for almost any substance lacking toxicity data and can provide supporting rationale to justify the use of read-across data. A defensible surrogate should possess the same functional groups in a configuration having similar local steric hindrance; have similar electron withdrawing/electron adding character; possess no additional unique functional groups which have their own significant associated toxicities or which can react with other nearby functional groups; display similar physical properties such as molecular weight range, volatility, hydrophobicity/hydrophilicity, acidity/basicity, and stability in solution; exhibit similar anticipated degradation pathways which yield the same or similar chemical classes of metabolites generated by similar metabolic reaction mechanisms such as hydrolysis or enzymatic degradation; and have similar predicted oral bioavailability based on ADME considerations. Where appropriate, candidate Tolerable Intake (TI) values and toxicity profiles should be derived for two or more potential surrogates to capture the range of functional group differences, and the final TI should account for the most stringent LOAEL and adverse effects among all relevant candidates.
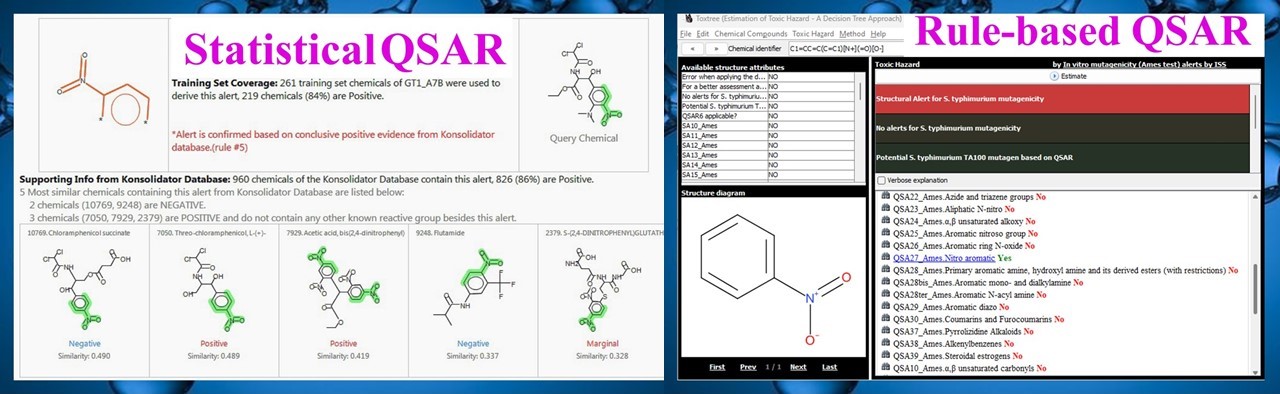
TDAC is a subject matter expert in Quantitative Structure Activity Relationship (QSAR) in silico predictions of mutagenicity, carcinogenicity, and sensitization potential, using software such as ToxTree, OECD Toolbox, MultiCASE, VEGA-QSAR, Lhasa Derek Nexus, and Sarah Nexus. For compounds that are not mutagenic or carcinogenic, in silico models are used to categorize non-cancer hazards into Cramer classes for compounds lacking published toxicity data.
Even for compounds that are genotoxic and likely to be carcinogenic, TDAC has been able to demonstrate with a high level of confidence that levels of extractable constituents detected in or on a medical device fall within the safe risk range. TDAC evaluates toxicological risks from exposure to potential carcinogens following accepted methodologies including the cancer slope factor, TD50, and threshold carcinogen models, which are facilitated by application of pragmatic and plausible less-than-lifetime exposure assumptions utilizing analytical data from exhaustive extractions.
TDAC’s principal toxicologist has contributed several key risk assessment methodology improvements that were adopted in ISO 10993-17:
- Added calculation methods for deriving an acceptable cancer risk dose (Slope Factor and TD50)
- Added percent bioavailability adjustment of oral NOAEL for route-to-route extrapolation instead of using a UF value
- Added citation of US EPA Carcinogen Guidelines to justify scenarios for prorating less-than-lifetime cancer risk
- Added caveat to avoid summing non-cancer risks for chemicals affecting a target organ if a mode of action is not evident
- Recommended adopting a revised default adult body weight of 60 kg instead of 50 kg (reduces overconservative bias)